Your organization deserves reliable identification
OUR BRANDS
World-class identification solutions for efficiency, safety, and compliance
PDC serves these diverse markets through a variety of channels by leveraging its combined resources of brands and experience across numerous industries to ensure superior product design, manufacturing and customer support.
THE LATEST FROM PDC
FROM THE PDC NEWSROOM
April 5, 2023
PRESS RELEASE
PDC Launches SecurBand® Wristbands to Provide Simple Visual ID for Events and Entertainment Venues
PDC, a global leader of identification solutions, announced today the launch of the SecurBand® wristband for single-day events. Developed to be the most affordable single-day-use wristband for simple and easy identification, SecurBand® wristbands are highly-vibrant and easy to see at a distance, making them the perfect choice for controlling event access. Their non-transferable strong adhesive and tamper-evident cuts prevent reuse, increasing security.
March 6, 2023
PRESS RELEASE
Vizient, Inc. Awards National Contract to PDC for Medical Labels
PDC, a global leader in healthcare identification and patient safety solutions, announced today that it has been awarded a contract with Vizient, Inc., the nation’s leading healthcare performance improvement company. The contract, effective March 1, 2023, gives Vizient members access to increased savings for the PDC medical label portfolio, which is the largest in the industry. It includes pharmacy labels, laboratory labels, chart labels, pre-printed communication labels, anesthesia labels, anesthesia tapes and more.
October 25, 2022
PRESS RELEASE
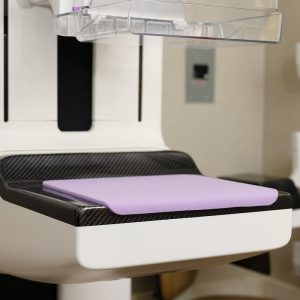
PDC Launches Comfort Cushion™ Mammography Pad to Help Breast Health Centers Improve the Patient Experience
PDC, a global leader of innovative identification and patient safety solutions, announced today the launch of the Comfort Cushion™ Mammography Pad for use by mammography providers, including hospitals and breast and women’s health centers. The cushy pad is applied by mammography technicians to the receptor plate of the mammography machine to reduce patient discomfort during compression. While October is Breast Cancer Awareness month, a time when the nation emphasizes the need for screening for early detection of cancer, mammography clinicians are concerned that mammography screening volumes still have not returned to pre-COVID-19 levels.
January 24, 2022
PRESS RELEASE
PDC announces a distribution agreement with Cancard, Inc. for the Canadian market
PDC has selected Cancard, Inc. as a distributor to offer PDC’s complete portfolio of identification solutions for patients, people, and products in Canada.
November 1, 2021
PRESS RELEASE
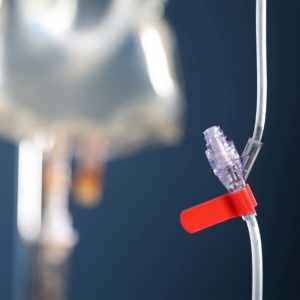
PDC Introduces Ident-Alert® IV Port Clips to Help Anesthesiologists Prevent Medication Errors
Colored Clips Developed by PDC and Anesthesiologist, Dr. Christopher Walters, Provide a Visual Cue at the IV Injection Port
“I love this company. ‘Fast and Efficient’ with great customer satisfaction!”
– Heather Eades
MARY WASHINGTON HOSPITAL
“I’ve been working with IDenticard for 12 years and I would never consider changing. Our patients and staff rely on us for security, and we rely on IDenticard.”
– SECURITY MANAGER AT A MAJOR MASSACHUSETTS HOSPITAL
“The guests love the quality and color variety of the wristbands, especially the kids. Kids love collecting the different color wristbands, to show off their fun memories with family and friends.”
– Ed Hodgdon
FUNTOWN SPLASHTOWN USA